Introduction:
Acute myeloid leukemia (AML) with myelodysplasia-related gene mutations is a new disease entity introduced in the International Consensus Classification of Myeloid Neoplasms 2022 (ICC 2022). Myelodysplasia-related gene mutations include ASXL1, BCOR, EZH2, RUNX1, SF3B1, SRSF2, STAG2, U2AF1, or ZRSR2 .
Allogeneic hematopoietic stem cell transplant (HCT) represents a curative option for subgroups of AML patients, but not for all. In a prior study (Song et al.) reported the survival benefit of allogeneic HCT in 59 patients with AML with secondary type mutations (STM) including SRSF2, SF3B1, U2AF1, ZRSR2, ASXL1, EZH2, BCOR, and STAG2. Allogeneic HCT could abrogate adverse prognosis in AML with STMs, and reduce its mortality risk by 58% (Song et al. BMT 2022).
In the present study, we aim to evaluate the frequency of this new disease entity of AML with MDS-related mutations and evaluate the prognostic impact of allogeneic HCT in patients with MDS-related mutations achieving CR1.
For comparison of the benefit from HCT, the mantel-Byar test was applied to take into account time-dependent covariate of HCT in CR1 to avoid immortal bias.
Patients and Methods:
We conducted a retrospective study that included 1,228 adult patients who were diagnosed with AML from January 1997 to January 2020and achieved CR1 in 5 centers (Toronto, Vancouver, and Montreal, Canada; Hwasun and Seoul, South Korea). Next-generation sequencing was performed as their standard clinical practice using targeted sequencing panel in each institution. As defined in ICC 2022, AML with myelodysplasia-related gene mutations was defined as the case of AML with ASXL1, BCOR, EZH2, RUNX1, SF3B1, SRSF2, STAG2, U2AF1, or ZRSR2 mutation(s). We considered allogeneic HCT in CR1 as a time-dependent covariate and used a Mantel-Byar test to avoid immortal bias. We identified 278 pts (22.6%) who had at least one MDS-related gene mutation at diagnosis. Relapse-free survival (RFS) at 2 years was selected for the primary endpoint, defined as the time from CR1 to relapse or death from any cause. EZR software version 1.41 was used throughout the study for statistical analysis.
Results:
The median follow-up duration among survivors after achieving CR1 was 16.5 months (mo) (Range: 1.3-217.9 mo). Between the HCT vs non-HCT group, there were no differences in sex, cytogenetic risk or other somatic mutation except NPM1 mutation which was more frequently detected in non-HCT group (p <0.001). As expected, younger pts were more frequently noted in HCT group, p=0.001).
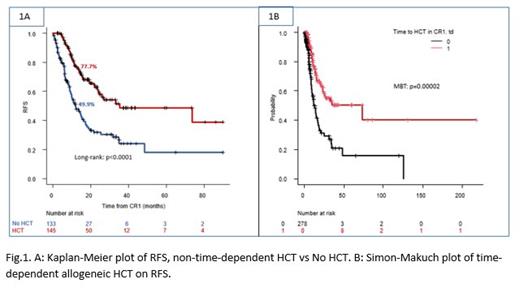
First, considering HCT as non-time dependent covariate, we have compared RFS and OS between HCT vs non-HCT groups. The 1 year RFS rate was superior in the HCT group (77.7%) to that in non-HCT group (49.9%, p<0.0001; Fig.1A). A similar trend was noted for the 1-year OS rate presenting higher OS rate in the HCT group (83.8%) than in the non-HCT group (72.7%, p=0.0003).
Next, when HCT in CR1 was taken into account as a time-dependent (td) covariate, Mantel-Byar analysis showed a consistent finding of superior RFS in HCT group to non-HCT group: time dependent covariate of HCT in CR1 was found to be favorable (Hazard ratio [(HR) of 0.45, (95% C.I. [0.31-0.65]; p=0.00003)] as shown in Fig.1B which is a Simon-Makuch plot.
In multivariate analysis (MVA) for RFS, time-dependent covariate of HCT in CR1 retained its statistical significance, thus concluding that HCT in CR1 is an independent prognostic factor for RFS (HR: 0.44, 95% CI [0.29-0.68], p =0.0002) together with other variables found to be adverse on RFS including monosomal karyotype (p=0.03), IDH1 mutation (p=0.01), and age > 60 years (p=0.002); while CEBPA (p=0.02) and NPM1 (p=0.049) mutations were found to be favorable on RFS in MVA.
Conclusion:
In our cohort of 1228 AML patients, we have identified 278 AML patients with myelodysplasia-related gene mutations (22.6%). We have confirmed that allogeneic HCT in CR1 is strongly recommended in a subgroup of AML with myelodysplasia-related gene mutations based on the favorable impact of HCT in CR1 on RFS from the present study. This result supports the urgent referral of this patient group for allogeneic HCT in CR1.
Disclosures
Bergeron:Jazz: Honoraria; Gilead: Honoraria; Taiho: Honoraria; Abbvie: Honoraria; Pfizer: Honoraria; Amgen: Honoraria; BMS: Honoraria. Kim:BL & H: Research Funding; Abbvie: Consultancy, Honoraria; Astellas: Consultancy, Honoraria; AIS biosicienc: Consultancy, Honoraria; AML-Hub: Consultancy, Honoraria; Bristol Myers Squibb: Consultancy, Honoraria; Daiichi-Sankyo: Consultancy, Honoraria; Boryung Pharm Co.: Consultancy, Honoraria; Amgen: Consultancy, Honoraria; Janssen: Consultancy, Honoraria; Pfizer: Consultancy, Honoraria; Novartis: Consultancy, Honoraria; Meiji Pharm: Consultancy, Honoraria; Sanofi: Consultancy, Honoraria; Takeda: Consultancy, Honoraria; Greencross Pharm: Consultancy, Honoraria; LG Chem: Consultancy, Honoraria. Sanford:Astellas: Honoraria; AbbVie: Honoraria. Schuh:Pfizer: Consultancy, Honoraria; Servier: Honoraria, Research Funding; Teva: Consultancy, Honoraria; Kite/Gilead: Research Funding; Glycomimetics: Research Funding; Bristol Myers Squibb: Honoraria, Research Funding; Astellas: Honoraria, Research Funding; Amgen: Honoraria, Research Funding; Agios: Honoraria, Research Funding; Abbvie: Honoraria, Research Funding. Kim:BMS: Research Funding; Pfizer: Honoraria, Research Funding; Paladin: Research Funding; Novartis: Consultancy, Honoraria, Research Funding.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal